 |
.
|
| |
How is PD Treated?
Currently, there is no cure for Parkinson’s disease. Instead,
therapy is directed at treating the symptoms that are most
bothersome to an individual with Parkinson’s disease. For this
reason, there is no standard or “best” treatment for Parkinson’s
disease that applies to every patient. NPF is currently
gathering the data to develop an individualized best approach to
patient care. Treatment approaches include medication
and surgical therapy. Other treatment approaches include
general lifestyle modifications (rest and exercise), physical
therapy, support groups, occupational therapy and speech
therapy. In this section, you will become more familiar with the
different types of medications commonly prescribed for PD, other
alternative therapies, and surgical treatment options. Recent
studies have implicated that a treatment is better than no
treatment. In other words, medications and therapies may modify
the progression of Parkinson’s disease.
|
Medications for Motor Symptoms
Since most symptoms of PD are caused by the lack of
dopamine
in the brain, many Parkinson’s drugs are aimed at either temporarily
replenishing dopamine or mimic the action of dopamine. These types
of drugs are called dopaminergic. These medications generally help
reduce muscle
rigidity, improve speed and coordination of movement and
lessen tremor.
Always remember that medication usage is only part of the whole
treatment plan for effectively treating PD. Learn more about the
available medications for the treatment of PD now.
Caution: PD medications may have interactions
with certain foods, other medications, vitamins, herbal supplements,
over the counter cold pills and other remedies. Anyone taking a PD
medication should talk to their doctor and pharmacist about
potential drug interactions.
Keep track of your medications using this customizable
Medication Schedule (Excel format).
Surgical Treatment Options
While surgery can be an effective treatment option for different
symptoms of PD, only the symptoms that previously improved on
levodopa have the potential to improve after the surgery. Surgical
treatment is reserved for PD patients who have exhausted medical
treatment of PD
tremor or who suffer profound motor fluctuations (wearing off
and dyskinesias). Learn more now about the available surgical
treatment options and visit the
Ask the Surgical Team forum, where Kelly D. Foote, M.D. and
Michael Okun, M.D. answer questions about surgical matters and
Parkinson's
disease.
Deep Brain Stimulation
 What are
the Facts? What are
the Facts?
- Deep brain
stimulation (DBS)
is a surgical procedure used to treat a variety of disabling
neurological symptoms—most commonly the debilitating symptoms of
Parkinson’s disease (PD), such as
tremor,
rigidity,
stiffness, slowed movement, and walking problems.
- The procedure is also used to treat
essential
tremor, a common neurological movement disorder.
- DBS does not damage healthy brain tissue by destroying nerve
cells. Instead the procedure blocks electrical signals from
targeted areas in the brain.
- At present, the procedure is used only for patients whose
symptoms cannot be adequately controlled with medications.DBS uses
a surgically implanted, battery-operated medical device called a
neurostimulator—similar to a heart pacemaker and
approximately the size of a stopwatch—to deliver electrical
stimulation to targeted areas in the brain that control movement,
blocking the abnormal nerve signals that cause tremor and PD
symptoms.Before the procedure, a neurosurgeon uses magnetic
resonance imaging (MRI)
or computed tomography (CT) scanning to identify and locate the
exact target within the brain where electrical nerve signals
generate the PD symptoms.
- Some surgeons may use microelectrode recording—which involves
a small wire that monitors the activity of nerve cells in the
target area—to more specifically identify the precise brain target
that will be stimulated.
- Generally, these targets are the
thalamus,
subthalamic
nucleus, and
globus
pallidus.
- Once the system is in place, electrical impulses are sent from
the neurostimulator up along the extension wire and the lead and
into the brain. These impulses interfere with and block the
electrical signals that cause PD symptoms.
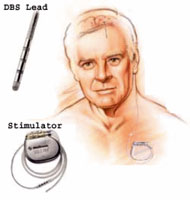 The DBS system
consists of three components: The DBS system
consists of three components:
- The lead- (also called an electrode)—a thin, insulated wire—is
inserted through a small opening in the skull and implanted in the
brain. The tip of the electrode is positioned within the targeted
brain area.
- The extension- is an insulated wire that is passed under the
skin of the head, neck, and shoulder, connecting the lead to the
neurostimulator.
- The neurostimulator- (the "battery pack") is the third
component and is usually implanted under the skin near the
collarbone. In some cases it may be implanted lower in the chest
or under the skin over the abdomen.
-
here are many brain targets that the DBS lead may be
placed
within; which one should you choose?
- There are three brain targets that have been FDA approved for
use in Parkinson’s disease.
- The most commonly utilized brain targets include the
subthalamic nucleus (STN) and also the globus pallidus interna (GPi).
- Target choice should be tailored to a patient’s individual
needs (Benabid, Benazzouz et al. 1998; Okun and Vitek 2004;
Benabid, Chabardes et al. 2005; Rodriguez, Fernandez et al. 2007).
- There are many ongoing studies that will help to refine target
choice for individual patients.
- Although the picture is not yet clear on the issue of target
choice, the STN does seem to provide more medication reduction,
while GPi may be slightly safer for language and cognition.
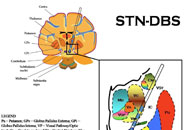
View pictorial representations of each of the three main DBS
targets used in Parkinson’s disease (figures were used with
permission of Dr. Okun).


What is the prognosis?
Although most patients still need to take medication after
undergoing DBS, many patients experience considerable reduction of
their PD symptoms and are able to greatly reduce their medications.
The amount of reduction varies from patient to patient but can be
considerably reduced in most patients. The reduction in dose of
medication leads to a significant improvement in side effects such
as dyskinesias (involuntary movements caused by long-term use of
levodopa). There is a 1-3% chance of infection,
stroke,
cranial bleeding, or other complications associated with anesthesia,
per side that is done. It is best to discuss the risks associated
with your neurologist because there are many risk factors, including
underlying medical conditions.

How do I know if I am a good candidate for DBS?
- You have had PD symptoms for at least five years.
- You have “on/off fluctuations, with or without
dyskinesia.
- You continue to have a good response to PD medications,
especially carbidopa/levdopa, although the duration of response
may be insufficient.
- You have tried different combinations of levodopa/carbidopa
and dopamine
agonists
under the supervision of a movement disorders neurologist.
- You have tried other PD medications, such as entacapone,
tolcapone, selegiline or amanadine without beneficial results.
- You have PD symptoms that interfere with daily activities.
- For more information, visit
DBS-STN.org,
an affiliate of the Parkinson Alliance, which is committed to
improving the quality of life for DBS-STN patients and their
caregivers.
Want to Learn More?
Medical content reviewed by: Nina Browner, MD—Medical Director of
the NPF Center of Excellence at the University of North Carolina at
Chapel Hill in North Carolina and by Fernando Pagan, MD—Medical
Director of the NPF Center of Excellence at Georgetown University
Hospital in Washington, D.C.

|
|
|